Special Report: The Evolution of Biomarker Use in Clinical Trials for Cancer Treatments
By Alex Vadas, Ph.D., T.J. Bilodeau, and Chintan Oza, Ph.D.
With an Introduction by Edward Abrahams
Note: Adapted from a report titled The Evolution of Biomarker Use in Clinical Trials for Cancer Treatments: Key Findings and Implications, published in October by the Personalized Medicine Coalition in collaboration with L.E.K. Consulting.
INTRODUCTION
By Edward Abrahams
When the Personalized Medicine Coalition first published The Case for Personalized Medicine in 2006, the pharmaceutical industry was skeptical that it could or should develop drugs for subpopulations based on identifiable biomarkers. Outside of a small community of molecular biologists steeped in the knowledge of pharmacogenomics, a field that suggested that scientists could identify which patients a drug would work for, personalized medicine was a daunting concept, especially to those in the industry with a more commercial focus.
The industry’s skepticism derived both from a then-limited appreciation of human heterogeneity at the molecular level (especially in tumors) as well as a business model that assumed that the industry should only develop drugs for “all comers,” even if they only worked for subsets of the public — and that companies could sell these drugs for relatively low prices.
That challenge was compounded by an unclear regulatory system that inhibited integrating diagnostics into health care; by an overtaxed reimbursement system that was slow to comprehend the value of diagnostics,; the necessarily higher prices for targeted drugs for smaller populations; and by the fact that most health care providers were trained before the human genome was mapped. These factors limited their full appreciation of the new science and technologies that underpinned personalized medicine and, in turn, their willingness to make the necessary changes to integrate it into health care.
Suffice it to say that the business, policy, and educational challenges have not disappeared and will get worse if price controls based on population averages are put in place. But the industry’s skepticism has, especially in oncology, all but been eradicated as it has embraced the principles of personalized medicine.
In 2005, personalized medicines, which PMC defines as drugs that include biomarker strategies in their respective labels, represented only five percent of the new molecular entities approved by the U.S. Food and Drug Administration. Last year, the figure was 42 percent, with most of the new approvals coming in oncology with enormous significance for the future of cancer care.
In this report, Alex Vadas, Ph.D., T.J. Bilodeau, and Chintan Oza, Ph.D. (in the Life Sciences & Pharma practice at L.E.K. Consulting) document the industry’s increasing commitment to developing personalized medicines in oncology. Whereas previous analyses of the pharmaceutical pipeline, including the one PMC commissioned five years ago, relied on impressionistic survey data, this more definitive study analyzes the Aggregate Analysis of Clinical Trials, a cloud-based platform on which all clinical trials are registered.
This accelerating shift in drug development has profound implications for key stakeholders across the health care spectrum, including the pharmaceutical and diagnostic industries, providers, payers, and, most importantly, patients. Benefits will include identification of responders/non-responders and development of drugs for responders that can be made safer, more effective, and, over time, potentially less expensive for systems that incorporate personalized medicine.
As a result, patients will have better outcomes due to earlier detection and more effective treatments.
Edward Abrahams, Ph.D., is president of the Personalized Medicine Coalition, a nonprofit education and advocacy organization in Washington, DC.
ANALYSIS OF KEY FINDINGS & IMPLICATIONS
By Alex Vadas, Ph.D., T.J. Bilodeau, and Chintan Oza, Ph.D.
To generate critical insights on macro biomarker trends, we explored the entirety of oncology clinical trials registered on ClinicalTrials.gov using a combination of automated analytical techniques and manual curation. We focused first on searching the AACT database (~281,500 clinical trials) for ~2,500 oncology drugs from 2000 to 2018 for the inclusion of biomarkers.
A defined list of biomarkers (including various synonyms) was created based on a variety of input sources to query the trials.1 This yielded ~11,000 oncology clinical trials using biomarkers for any purpose. These ~11,000 clinical trials were analyzed to gain useful insights, including macro trends in biomarker use over time; biomarker use by tumor type; utilization trends for specific biomarkers; number of biomarkers used per trial; trial sponsor; and several other findings, many of which are discussed in this report.
Findings
Based on our analysis, we observed a growing use of biomarkers in clinical trials with ~55 percent of all oncology clinical trials in 2018 involved the use of biomarkers, as compared with ~15 percent in 2000. Our study included all oncology therapy trials using chemotherapies and non-targeted therapies, which are less likely to use biomarker-focused approaches.
While there has been a ~9 percent compound annual growth rate (CAGR) for total oncology clinical trials since 2000, trials involving biomarkers grew at nearly twice this rate, at ~17 percent CAGR, during the same period (Figure 1). This study focused exclusively on oncology clinical trials; however, a similar trend toward increased use of biomarkers has been observed across other therapeutic areas (data not shown).
Recent advancements in tumor biology, assay techniques, and tumor profiling solutions have influenced clinical trial design, especially in terms of the number of biomarkers tested in a trial. Almost half of the total trials with biomarkers specifically highlighted interrogation of two or more biomarkers in 2018, compared with just ~14 percent in 2000 (Figure 1).
FIGURE 1. THE NUMBER OF ONCOLOGY TRIALS WITH BIOMARKERS GREW AT TWICE THE RATE OF ONCOLOGY TRIALS OVERALL FROM 2000 – 2018, AS MEASURED BY COMPOUND ANNUAL GROWTH RATE (CAGR)
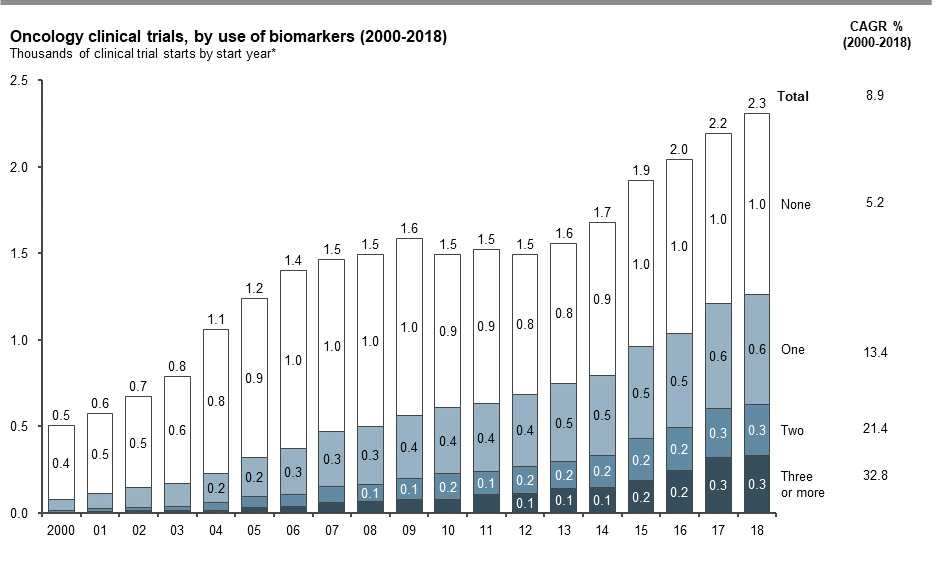
Note: * Number of biomarkers for trials that include both unspecified and specified biomarkers are counted as per the number of specified biomarkers; trials with only unspecified biomarkers are counted under one biomarker.
Sources: AACT; L.E.K. biomarker database
Overall, biomarkers are being explored across all cancers, ranging from 25 percent to 60 percent of trials from 2000 to 2018; use is highest in breast, leukemia, lung, lymphoma, prostate, melanoma, and head and neck cancers. While growth in biomarker trials since 2000 has been significant across most cancers, lung has been notably outpacing others, doubling the biomarker trial count in the past five years alone (Figure 2).
FIGURE 2. GROWTH IN BIOMARKER TRIALS SINCE 2000 HAS BEEN SIGNIFICANT ACROSS MOST CANCERS
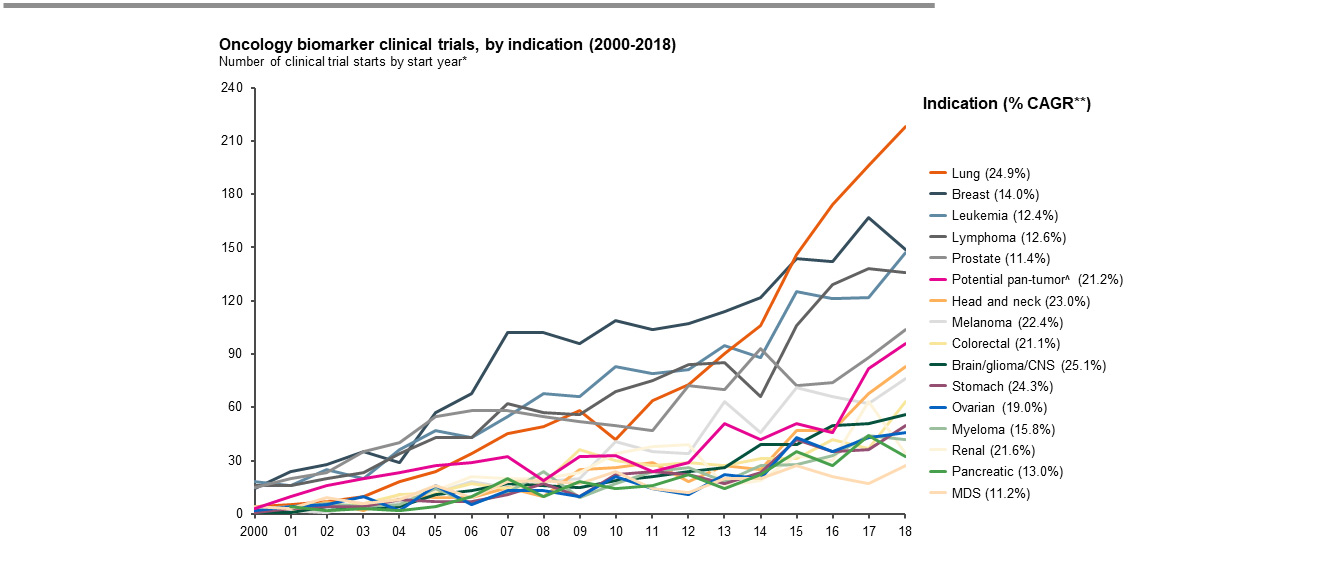
Note: * Trials looking at two or more indications are repeat counted for each indication; ** % indicates CAGR 2000 – 2018; ^ Potential pan-tumor biomarker trials were determined based on a proxy of trial activity that could potentially lead to a pan-tumor label and included oncology biomarker clinical trials in Phase 2, 3, or 4 with > five specified tumors per trial or < five tumors per trial but including a general indication term (e.g., cancer, malignancies, metastasis/metastases, oncology, neoplasm, tumour/tumor)
Biomarkers tested in clinical trials are driven by the mechanisms targeted by precision therapies and immunotherapies, including prognostic and monitoring biomarkers. For example, HER2, EGFR, KRAS, BRCA1/2, and PD1/PD-L1 are biomarkers closely linked to therapeutic mechanisms in cancers such as breast cancer, non-small cell lung cancer, and ovarian cancer. MRD and PSA are key biomarkers for monitoring in heme and prostate cancers, respectively.
Significant growth has also been seen in trials exploring pan-tumor biomarkers including MSI and NTRK, both of which are associated with pan-tumor indication approvals – MSI associated with pembrolizumab and NTRK with larotrectinib and entrectinib. For example, in the past five years, there were 31 trials conducted for the NTRK gene fusion biomarker and 63 trials for MSI.
Other potential pan-tumor biomarkers, including TMB and FGFR, are in trials that could result in future pan-tumor indications. These trends clearly reflect advances in the use of biomarkers to guide personalized drug development independent of the more traditional organ-specific clinical trial approaches. Ultimately (and irrespective of the rationale), biomarker testing has become a critical component in cancer research, clinical development, and treatment management.
Seventeen organizations, including five academic centers, have sponsored or co-sponsored 100-plus oncology trials involving a biomarker. Unsurprisingly, the National Cancer Institute, with dedicated research support to investigate biomarkers for targeted therapies along with a wide network of research laboratories and data centers, is leading the way in collaborating on or sponsoring oncology biomarker trials throughout the nation (see e.g. , The Biomarkers Consortium2). The biopharmaceutical companies that pursued the most biomarker trials between 2000 – 2018 are Roche/Genentech, Novartis, AstraZeneca/MedImmune, Bristol-Myers Squibb/Celgene, Merck, and Pfizer. Their focus on biomarkers during this time period was likely driven by their targeted therapy and immunotherapy portfolios and disease area focus.
Conclusion
Biomarkers have become highly valuable in driving oncology research as well as development and commercialization of targeted cancer therapies; we foresee continuing significant impact for all stakeholders across the health care continuum. In oncology specifically, the posture toward biomarker development for innovative oncology therapies is becoming the norm versus the exception, and the industry will need to adapt to this evolving paradigm of personalized medicine.
Biopharmaceutical companies have incorporated biomarker strategies to drive their research and commercial operations. Biomarkers are used to optimize their targeted therapy portfolios; make clinical development decisions; access and preserve bio-specimens; build and mine scaled clinical genomics datasets; segment commercial markets; develop market access and pricing strategies for biomarker-driven targeted therapies; and pursue broader collaborations with academic and diagnostic partners.
It will be critical for health care providers to educate patients and caregivers, prioritize precision medicine capabilities, including biomarker profiling as well as data management and interpretation infrastructure to enable precision medicine care delivery.
Diagnostic companies and clinical laboratories will play a major role in actively collaborating with biopharmaceutical companies and providers on FDA-approved assays as well as laboratory developed tests (LDTs). Multiplexing, sample-sparing, and liquid biopsy techniques that enable broader testing from smaller amounts of tissue or plasma samples may play a prominent role.
Regulators will need to provide comprehensive guidance on sample quality and testing, further simplify the approval process for companion diagnostics, and standardize guidance around the use of personal data for research purposes. In turn, payers and diagnostics developers will need to work with CMC to drive broader coverage and reimbursement of biomarker testing (see, e.g., Decision Memo for Next Generation Sequencing (NGS) for Medicare Beneficiaries with Advanced Cancer (CAG-00450N)3.)
As patients learn more about these capabilities, they may start to request advanced testing and ask their providers to use sample-sparing and lower-risk sampling approaches where possible and may contribute their de-identified data for clinical research. Examples for the latter would be liquid biopsies for tumors in lungs and the pancreas.
Ultimately, biomarker-driven personalized medicine approaches will lead to better patient outcomes by enabling early detection; identifying treatment responders; and monitoring treatment, response, and targeted therapeutic effect. Further, we anticipate these methods will expand to other therapeutic areas. For the immediate future, all signs point to cancer care will be profoundly influenced by biomarkers to guide researchers and physicians at every stage, from drug development to disease management.
1 We needed to query for the same biomarker using a range of synonyms (e.g., HER2, HER-2/neu, and ERBB2).
2 https://fnih.org/what-we-do/biomarkers-consortium
3 https://www.cms.gov/medicare-coverage-database/details/nca-decision-memo.aspx?NCAId=290
Sources: AACT; L.E.K. biomarker database
Alex Vadas, Ph.D., is a Managing Director in L.E.K. Consulting’s biopharmaceutical and life sciences practice and leads the firm’s diagnostics, research tools, and personalized medicine practice.
T.J. Bilodeau is a Principal in L.E.K. Consulting’s biopharmaceutical and life sciences practice and Director of the L.E.K. Healthcare Insights Center.
Chintan Oza, Ph.D., is a Senior Consultant in L.E.K. Consulting’s biopharmaceutical and life sciences practice.




